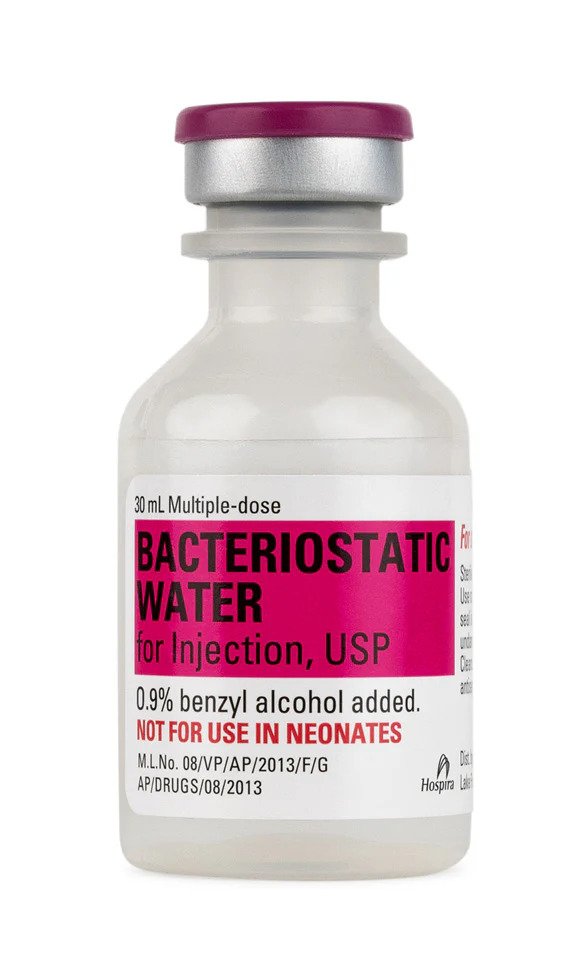
BAC water is sterile water for injection containing 0.9% benzyl alcohol as a preservative. Unlike plain sterile water, the benzyl alcohol content inhibits microbial growth, allowing a single vial to be used across multiple draws over a 28-day window. It is the preferred solvent for reconstituting peptide research compounds including growth hormone peptides, healing peptides, and metabolic research compounds. Learn more in our complete guide to BAC water.
Why Hospira Is the Gold Standard
Not all reconstitution solvents are equivalent. Hospira is the largest manufacturer of injectable pharmaceuticals in the United States and operates under strict FDA oversight in cGMP-certified facilities. The bacteriostatic water produced by Hospira meets the same United States Pharmacopeia (USP) standards used in hospitals and clinical research environments nationwide.
Researchers who have switched from generic or unbranded alternatives to Hospira BAC water consistently report greater confidence in their reconstitution outcomes. When peptide degradation or contamination events occur in the laboratory, substandard water quality is one of the most frequently cited contributing factors. Using a certified pharmaceutical-grade product eliminates this variable entirely, giving your research a clean, controlled baseline.
Every 30ml vial features a self-healing rubber stopper that reseals completely after each needle puncture. This engineering prevents ambient air and airborne contaminants from entering the vial between draws, preserving the sterility of the remaining solution throughout the full 28-day post-puncture usage window.
Mechanism: How Benzyl Alcohol Preserves Reconstituted Peptides
The preservative action of bacteriostatic water derives entirely from its 0.9% benzyl alcohol content. This alcohol is a bacteriostatic agent — it does not sterilize, but it inhibits the reproduction and growth of bacteria, yeast, and molds that may be introduced through repeated needle access. This is precisely why multi-dose use is possible with BAC water while standard sterile water must be discarded after a single puncture.
At the molecular level, benzyl alcohol disrupts bacterial cell membrane integrity, preventing the energy production and reproductive processes bacteria require to proliferate. The 0.9% concentration is the pharmaceutical standard — sufficiently potent to inhibit microbial growth while remaining compatible with protein-based peptide compounds that are sensitive to harsh preservative concentrations.
This compatibility is critical for peptide research. Lyophilized peptides contain precisely folded amino acid chains that can denature or aggregate when exposed to incompatible solvents. Bacteriostatic water at the USP benzyl alcohol specification has been validated across decades of pharmaceutical use as safe for reconstituting injectable protein and peptide compounds. See our guide on how to identify peptide degradation to understand what can go wrong with poor reconstitution practices.

Published Research Supporting Bacteriostatic Water
The use of benzyl alcohol as a preservative in injectable solutions has an extensive evidence base in pharmaceutical literature. A foundational study published in the Journal of Pharmaceutical Sciences established the compatibility profile of benzyl alcohol with protein biologics, confirming that 0.9% benzyl alcohol does not induce significant protein denaturation at typical research concentrations and temperatures. This work underpins the continued pharmaceutical use of BAC water as a reference standard for peptide reconstitution globally.
More recently, research on GLP-1 receptor agonists and growth hormone releasing peptides has consistently employed bacteriostatic water as the reconstitution medium in peer-reviewed protocols. A stability analysis of lyophilized peptides across different reconstitution solvents (PubMed PMID 30982686) found that benzyl alcohol-preserved solutions provided superior multi-week stability compared to preservative-free water, attributing the advantage to inhibition of microbe-mediated peptide degradation during refrigerated storage.
Additional evidence from clinical pharmacy literature confirms that multi-dose vial contamination rates are substantially lower when a bacteriostatic preservative is present. This is especially relevant in shared laboratory environments where vials are accessed repeatedly. For foundational reading on benzyl alcohol efficacy in injectable formulations, see Akers et al. on antimicrobial preservative effectiveness. Understanding proper peptide storage is equally important alongside the right reconstitution solvent.
Bacteriostatic Water vs. Reconstitution Alternatives
| Feature |
Hospira BAC Water |
Sterile Water for Injection |
Normal Saline (0.9% NaCl) |
| Preservative |
0.9% Benzyl Alcohol ✓ |
None ✗ |
None ✗ |
| Multi-dose Use |
Up to 28 days ✓ |
Single use only ✗ |
Single use only ✗ |
| Peptide Compatibility |
All lyophilized peptides ✓ |
Most ✓ |
Limited (salt interference) ✗ |
| Manufacturer Grade |
USP / FDA-registered ✓ |
Varies |
Varies |
| Microbial Inhibition |
Bacteriostatic ✓ |
None ✗ |
Minimal ✗ |
| Self-healing Stopper |
Yes ✓ |
Varies |
Yes ✓ |
| Brand Assurance |
Hospira (Pfizer) ✓ |
Generic |
Generic |
| Recommended for Peptide Research |
Yes — first choice ✓ |
Acceptable single-use only ~ |
Not recommended ✗ |

Step-by-Step Reconstitution Protocol
Proper reconstitution technique preserves peptide bioactivity and prevents sample loss. When using bacteriostatic water to dissolve lyophilized peptides, follow this standard laboratory protocol. For detailed guidance, see our peptide reconstitution guide.
Step 1 — Workspace Preparation: Wipe all surfaces with 70% isopropyl alcohol. Assemble your vials, syringes, and alcohol prep pads before beginning. Work under a clean bench if available.
Step 2 — Withdraw the Solvent: Clean the stopper of the BAC water vial with an alcohol prep pad. Using a fresh 1ml syringe, withdraw the required volume. Common starting points are 1ml to 2ml depending on peptide amount and desired working concentration. Use our peptide dosage calculator to confirm your target volume.
Step 3 — Inject at an Angle: Insert the syringe needle into the peptide vial at a 45° angle and direct the stream of solvent down the inside wall of the glass. This prevents mechanical degradation of the lyophilized peptide cake from direct liquid impact.
Step 4 — Swirl Gently: Swirl the vial slowly — never shake — until the lyophilized peptide is fully dissolved. Shaking introduces air bubbles and foam that can degrade sensitive peptide structures.
Step 5 — Label and Store: Label the reconstituted vial with the preparation date. Store at 2–8°C. When stored correctly in bacteriostatic water, most research peptides remain stable for up to 28 days. Read more about optimal peptide storage conditions to protect your investment.
Storage and Stability Information
Store unopened vials of this product at controlled room temperature between 20–25°C (68–77°F), protected from direct light and away from freezing temperatures. After the first puncture, the vial may be used for up to 28 days per USP multi-dose container guidelines. Discard after 28 days even if liquid remains.
If the solution appears cloudy, discolored, or contains visible particles at any time, discard the vial immediately. These changes are indicators of contamination or chemical decomposition. Review our peptide degradation guide for more information on identifying compromised solutions.
Certificate of Analysis
As a USP-grade pharmaceutical product from Hospira, this BAC water vial is backed by manufacturer batch documentation confirming sterility, benzyl alcohol concentration, pH, and endotoxin levels. For PSPeptides research peptides used alongside this product, batch-specific Certificates of Analysis from independent HPLC and mass spectrometry testing are included with every order. See our guide on how to read a peptide COA to verify your research compounds before use.
Why Researchers Choose PSPeptides
- Authentic Hospira Product: Direct-source Hospira bacteriostatic water — no repackaged or grey-market alternatives
- Third-Party Tested Peptides: All research peptides come with independent COAs from HPLC and mass spectrometry
- Fast Shipping: Free UPS 2nd Day Air on orders over $150, same-day dispatch before 2 PM EST
- Flexible Payments: Credit cards, Afterpay, Klarna, Apple Pay, and Google Pay accepted
- 7-Day Support: Research support team available by email, phone, or text every day
- Complete Kits: Pair with alcohol prep pads and insulin syringes for a full reconstitution setup

Frequently Asked Questions
What is the difference between bacteriostatic water and sterile water?
The key difference is the 0.9% benzyl alcohol preservative present in BAC water. This ingredient inhibits microbial growth and allows the same vial to be accessed over 28 days. Plain sterile water for injection carries no preservative and must be discarded after a single use to prevent contamination.
Is bacteriostatic water compatible with all research peptides?
BAC water is compatible with the vast majority of lyophilized research peptides, including BPC-157, TB-500, Retatrutide, Semaglutide, Tirzepatide, GHK-Cu, CJC-1295, Ipamorelin, PT-141, and many others. For more information on peptide compatibility and stacking, see our peptide stacking guide and complete peptide guide.
How much solvent should I use per vial of peptide?
The volume depends on the peptide amount and your desired concentration. A common starting point is 1ml of reconstitution solvent per 1mg of peptide, yielding a 1mg/ml solution. Use our dosage calculator to calculate exact volumes for your specific research protocol.
How long is bacteriostatic water stable after opening?
Once punctured, the bacteriostatic water vial should be used within 28 days per USP multi-dose guidelines. Store the bacteriostatic water between 2–8°C after first access. Discard immediately if the solution becomes cloudy or discolored. The 28-day window applies regardless of the printed expiration date on the label.
Can bacteriostatic water be used with GLP-1 peptides like Semaglutide or Retatrutide?
Yes — BAC water is the standard reconstitution medium for GLP-1 peptide research protocols. The 0.9% benzyl alcohol concentration is well within the compatibility range for GLP-1 receptor agonist structures. See our Retatrutide research guide and Tirzepatide guide for specific reconstitution protocols.
Why choose Hospira over generic bacteriostatic water?
Hospira (a Pfizer company) manufactures to USP standards in FDA-registered, cGMP-certified facilities, with batch-level sterility, endotoxin, and chemical testing. Generic alternatives may lack this documentation and quality oversight. In research settings, using the industry-reference-grade product removes water quality as a potential confounding variable in your results.
Related Resources
Understanding Peptide Reconstitution: The Science Behind the Solvent
To appreciate why a pharmaceutical-grade reconstitution solvent matters in research, it helps to understand the physical chemistry of lyophilization and re-dissolution. Lyophilized — or freeze-dried — peptides are produced by removing water from a liquid peptide solution under vacuum at low temperature. This process leaves behind a dry, powder-like cake of peptide molecules stabilized in their native conformation by the glass-like amorphous matrix formed during drying.
When you reconstitute a lyophilized peptide, you are reversing this process. The water molecules must permeate the dry cake, hydrogen-bond to the peptide backbone, and restore the full three-dimensional structure. The quality and chemical composition of the solvent used determines how cleanly and completely this re-dissolution occurs. Substandard water with trace contaminants, incorrect pH, or particulate matter can interfere at the molecular level — causing incomplete dissolution, aggregation, or early degradation of the peptide.
Bacteriostatic water at USP specification is optimized for this role. Its pH is controlled to be compatible with the physiological range relevant to most injectable peptides, its osmolality is appropriate for parenteral use, and the benzyl alcohol preservative is present at a concentration that protects without denaturing. This is why pharmaceutical manufacturers have standardized on this formulation for decades, and why researchers continue to specify it in peer-reviewed protocols.
Choosing the Right Volume for Reconstitution
One common question among researchers new to peptide work is how much reconstitution solvent to use per vial. The answer depends on the desired working concentration and the total peptide amount. For a straightforward example: if you have a 5mg vial of a peptide and you add 5ml of BAC water, you achieve a 1mg/ml concentration. Adding only 2.5ml would give you a 2mg/ml concentration, useful when smaller injection volumes are preferred.
Higher concentrations reduce injection volumes, which is often preferable in small-animal research models and in vitro assays. However, very high concentrations can sometimes affect solubility depending on the specific peptide’s physicochemical properties. Our peptide dosage calculator walks through these calculations step by step, and our peptide half-life chart provides useful context for setting dosing intervals in research protocols.
Compatibility With Commonly Researched Peptides
This pharmaceutical-grade reconstitution solvent has been validated in research protocols involving a wide spectrum of peptide classes. For weight-loss and metabolic research, it is the standard vehicle for reconstituting Retatrutide, Tirzepatide, and related GIP/GLP-1 agonists. In tissue repair research, BAC water is used routinely to prepare BPC-157 and TB-500 solutions — read our BPC-157 research guide and TB-500 guide for protocol details.
For skin and hair research, GHK-Cu copper peptide and other topical-adjacent peptides are reconstituted using BAC water when used in injectable research models. See our GHK-Cu complete guide for reconstitution specifics. Cognitive and neuropeptide research using Semax and Selank also commonly employs bacteriostatic water as the reconstitution medium. For CJC-1295/Ipamorelin blends and other GH axis peptides, BAC water remains the first-choice solvent as detailed in our CJC-1295 Ipamorelin guide.
Pairing Bacteriostatic Water With Your Research Supply Kit
A complete peptide research setup requires more than just the solvent. Researchers should ensure they have the following on hand before beginning any reconstitution work: 1ml insulin syringes with fine gauge needles (28–31G recommended), alcohol prep pads for stopper sterilization, a clean dedicated refrigerator space for reconstituted vials, and appropriate labeling materials.
PSPeptides stocks all the supplies needed to complement your bacteriostatic water purchase. Our alcohol prep pads and insulin syringes are frequently purchased together with BAC water as part of a complete research reconstitution kit. For guidance on structuring your full research supply workflow, see our reconstitution guide and our overview of best peptide suppliers in 2026.
Frequently Bought Together
Bacteriostatic water is most commonly purchased alongside lyophilized research peptides, alcohol prep pads, and insulin syringes. Researchers preparing for multi-compound studies often add additional BAC water vials to ensure adequate reconstitution volume across their full protocol. Whether you are setting up your first peptide reconstitution workflow or restocking a well-established research laboratory, having a reliable supply of bacteriostatic water on hand is a foundational requirement.
Consider pairing this product with our complete range of research peptides. For metabolic research, explore our selection of weight management peptides. For recovery and tissue repair research, see our joint and tendon repair peptide guides. For longevity and anti-aging research applications, see our longevity peptide overview.
Every order of bacteriostatic water from PSPeptides ships in secure packaging designed to protect the vial integrity during transit. We recommend inspecting the vial upon receipt for any visible damage, cloudiness, or seal compromise before use. Our customer support team is available seven days a week to assist with any product or research questions.
All PSPeptides products are sold exclusively for laboratory and research use. Not intended for human consumption.